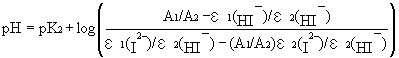A. Cruise narrative
a. Expedition
b. Chief Scientist
c. Ship
d. Ports of Call
e. Cruise dates
a. Cruise track (Fig. A.1)
Digital pictures, courtesy of Andy Girard.
b. Station sampling
c. Underway sampling
towed VPR (Video Plankton Recorder), with planktonic taxonomic
type and abundance, temperature,
conductivity, fluorescence, light attenuation and PAR
yoyoing to 80 meters depth once or twice between CTD stations.
d. Floats and drifters
The R/V Revelle departed Pusan, Korea on June 24, 1999 at 1600 in good
weather and
returned on July 17.
This was the seventh leg of the Hahnaro (HNRO) expedition.
Generally calm to moderate seas throughout the cruise. Air temperature
was in the 16-22 C range. There was occasional rain.
Three separate sampling programs were aboard: CTD/rosette/chemistry,
bio-optics,
and VPR (Video Plankton Recorder).
The cruise leg covered the Korean and Japanese sectors of the Japan/East
Sea.
The purposes of the cruise leg were to map the water properties and
geostrophic
circulation
of the Japan/East Sea from top to bottom, the bio-optical properties,
and
the plankton distribution.
The water properties and circulation of the Russian sector were
measured in a companion cruise on the Khromov, following the Revelle
leg.
CTD/rosette station sampling was to the bottom at each of the 112
stations.
Most stations were separated by 10 to 30 nautical miles. The station
pattern covered most of the southern and eastern Japan/East Sea.
One station near Dok Do was abandoned because
the local Korean patrol was not aware of our clearance to work.
One extra station (113) to 800 m was made on the return to Pusan in
order
to test the CTD which will be the backup CTD on the Khromov. On most
stations, 24 samples were collected from top to bottom. Maximum bottle
spacing in the deep waers was 250 meters with some exceptions. Most
sampling in the upper waters was based on the many
features in the CTD salinity and oxygen and the transmissometer.
An altimeter on the CTD/rosette frame was used for the bottom approach
on most
stations. A pinger on the CTD/rosette frame was used for several
stations.
A lowered acoustic doppler current profiler was used on every station.
The VPR was towed between most station pairs except for the longer
steams between sections. On most days two separate casts for bio-optics
were made. At these stations, extra samples for bio-optical properties
were often collected from near-surface rosette bottles from the CTD
cast.
A plankton net tow was done at 15 stations.
Alkalinity and pH:
A comparison of alkalinity and pH methods between the Seoul National
University group under Kyung-Ryul Kim (Dong-Jin Kang aboard the
Revelle) and the Pacific Oceanological Institute group under Pavel
Tishchenko was carried out during the cruise. POI sampling for pH and
alkalinity was at every station. SNU sampling was at 15 stations for
comparison of methods. The results of the comparison are included in
section B.6.c.
CFC:
Samples for CFCs were collected in glass ampoules for analysis
at the UW laboratory and comparison with analyses carried out on the
Revelle. All CFC sampling on the Khromov will be using these glass
ampoules.
CTD fish numbers used:
The rosette consisted of:
CTD#3:
Bottles:
Thermometers:
Transmissometer:
Winches:
Station-Cast number assignments:
SALINOMETER TYPES SERIAL NUMBERS
WORMLEY standard water used:
Comments:
Autosals were configured for computer-aided measurement. The data
were
acquired on a PC.
#48-263 stations 1-113 24 deg bath temp
Oxygens were run on all stations using a Dosimat UV-endpoint detection
automatic titration system.
Comments:
Nutrients were measured on all stations using a Technicon AA-II CFA
system
with a PC based acquisition system. Nutrients measured - NO2, NO3, PO4,
SIO3.
Comments:
The system performed well with few problems. Data were reviewed by
analysts
and transferred to the processing computer for integration with
other water sample data.
epsilon1(I2-)/
epsilon2(HI-) = 2.222
epsilon2(I2-)/
epsilon2(HI-) = 0.133 All SNU data reported here are averaged
value of duplicate analysis. The average precision of duplicate analysis is
0.006 pH unit is one standard deviation. POI used potentiometric measurement
in a potential cell without liquid junction for pH measurements of
seawater, since it was reported
that unreproducibility and loss of accuracy of potentiometric pH
measurement are caused by liquid junction potential (Tishchenko and
Pavlova, 1999).
A.1. Highlights
HNRO7 (Expedition Hahnaro Leg 7)
Lynne D. Talley
Scripps Institution of Oceanography 0230
La Jolla, CA 92093-0230 USA
ltalley@ucsd.edu
R/V Revelle, Captain David Murline
Pusan, Korea
24 June 1999 - 17 July 1999
A.2. Cruise summary
List of events, from ship's
officers, with all station (CTD, optical, net tow) and VPR towing times.
CTD station locations and times
in WOCE Hydrographic Programme format.
113 CTD/24-bottle rosette stations; 112 stations included LADCP
(2156 bottles tripped)
Water sampling to the bottom for temperature, salinity, oxygen,
transmissometer,
nitrate, phosphate, silicate, nitrite, CFC's, pH,
alkalinity, C14, del18O, helium, tritium, argon, neon. Surface sampling
at
selected station locations for delta-C13, phytoplankton growth
rates and calcite. Average depth of cast: 2500 m.
37 Bio-optical casts
15 Net tows near the surface
pCO2
surface temperature and salinity
Seabeam center beam bathymetry
Knudsen echo sounder bathymetry
ADCP (Acoustic Doppler Current Profiling)
meteorology
2 Minimet surface drifters
2 Profiling ALACE floats ballasted to 800 meters
A.3. Narrative
A.4. Interlaboratory comparisons of chemistry methods
A.5. List of principal investigators
A.6. Cruise participants
Institution acronyms
B. Description of measurement techniques and calibration
B.1. CTD (conductivity-temperature-depth): Carl Mattson (SIO/ODF)
CTD data were recorded on IBM PC's.
Digital backups were made on CDROMS and Zip disks.
Analog backups were made on VCR cassettes.
NBIS Model MKIII ODF CTD#3 stations 1-8,113
NBIS Model MKIII ODF CTD#5 stations 9-112
NBIS MKIIIB CTD s/n 01-1095 (ODF ctd#3) sta 1-8, 113
Comments:
NBIS MKIIIB CTD s/n 01-1070 (ODF ctd#5) sta 9-112
Sensormedics Oxygen Sensor s/n 6-12-07 sta 1-108
Sensormedics Oxygen Sensor s/n 6-12-08 sta 109
Sensormedics Oxygen Sensor s/n 6-02-08 sta 110-113
FSI OTM s/n 1322 sta 113
STS 24 bottle rosette frame
24pl Seabird pylon model SBE32 s/n 3212613-0164
Seabird Temperature Sensor SBE35 s/n 3516590-0011
SIO made Bullister style 10 liter bottles
Benthos Pinger model 2216 s/n 1275
Simrad Altimeter model 807 s/n 0711090
STS Battery Pack for Altimeter
RDI LADCP CS-150KHZ s/n 1546
LADCP Battery Pack
Wetlabs Cstar 25cm transmissometer c/n CST-244DB
Wetlabs Cstar 25cm transmissometer c/n CST-245DB
Conductivity sensor failed during Sta 9 cast 1.
CTD#5:
Ctd#3 was replaced by CTD#5 prior to sta 9 cast 2.
FSI OTM #1322 was the second temp sensor on sta 113
The conductivity sensor drifted again on sta 113.
CTD #5 has dual sensors mounted on twin turrets -
two identical Temperature channels and two identical conductivity
channels.
CTD sensors soaked in distilled water between all casts.
Swapped sensor pair in config file starting sta 59.
PRT#2 and COND#2 were the most stable sensor pair so these were used in
onboard data processing operations for both CTD and bottle data
reports.
PRT#1 (after about sta92) was observed to jump about
0.0008 deg on casts greater that 3200M. It was usually
observed on the upcasts coming through about 3300M then
jumped back to overlap downcast trace when it
comes back up - around 3000M. Could be a digital
bit sticking in that channel (bit #5?).
Cond#1 sensor has a pressure effect on deep casts
and will require a pressure fit correction.
10L Bullister style, SIO manufactured.
Bottles serial numbered 1-24 corresponded to the pylon tripping
sequence 1-24 with the first bottle tripped being bottle #1.
Bottles serial numbered 1-24 were used on all casts.
The SBE35 Ref temp sensor data was recorded on all bottle trips.
CTD oxygen:
No DSRT's
Oxygen data interfaced with the CTD and incorporated into
the CTD data stream using a:
Sensormedics Oxygen Sensor s/n 6-12-07 sta 1-108
Sensormedics Oxygen Sensor s/n 6-12-08 sta 109
Sensormedics Oxygen Sensor s/n 6-02-08 sta 110-113
Wetlabs Cstar 25cm (Blue) Transmissometer c/n CST-244DB
Wetlabs Cstar 25cm (RED) Transmissometer c/n CST-245DB
Forward Markey CTD winch used on all casts
No wire or winch problems throughout the cruise.
Cast numbers were assigned between the CTD and the
Bio-Optical profiler depending on which was deployed first.
Station 9 was the only station that the CTD was deployed
on two casts.
B.2. Salinity analyses: Carl Mattson (SIO/ODF)
Guildline 8400A Autosal 55-503
Guildline 8400A Autosal 48-263
Batch P-134
203 vials used
2 bad vials
B.3. Oxygen water sample analyses: Carl Mattson and Ron Patrick
(SIO/ODF)
No major problems, hardly any problems.
The titrator employed a Brinkman Dosimat 665 automatic burette and
an
Ultraviolet detection system interfaced with a PC for data
acquisition
and control.
B.4. Nutrient analyses: Carl Mattson and Doug Masten (SIO/ODF)